
Available studies suggest limited efficacy of zolbetuximab monotherapy in 2L/3L and zolbetuximab + pembrolizumab in 3L for advanced G/GEJ adenocarcinoma
In Cohort 1A of the Phase 2 ILUSTRO trial (n= 30):1
In Cohort 3A of the Phase 2 ILUSTRO trial (n = 3):1
In the Phase 2 MONO trial (n = 54):2
In the Phase 2 FAST trial and the Phase 3 SPOTLIGHT and GLOW trials, patients who received previous chemotherapy for advanced G/GEJ adenocarcinoma were excluded.3–5 However, patients may have received previous perioperative chemotherapy as long as it was completed at least 6 months prior to randomization.
Table 1. Phase 2 clinical trials evaluating zolbetuximab as ≥ 2L treatment of advanced or metastatic G/GEJ adenocarcinoma
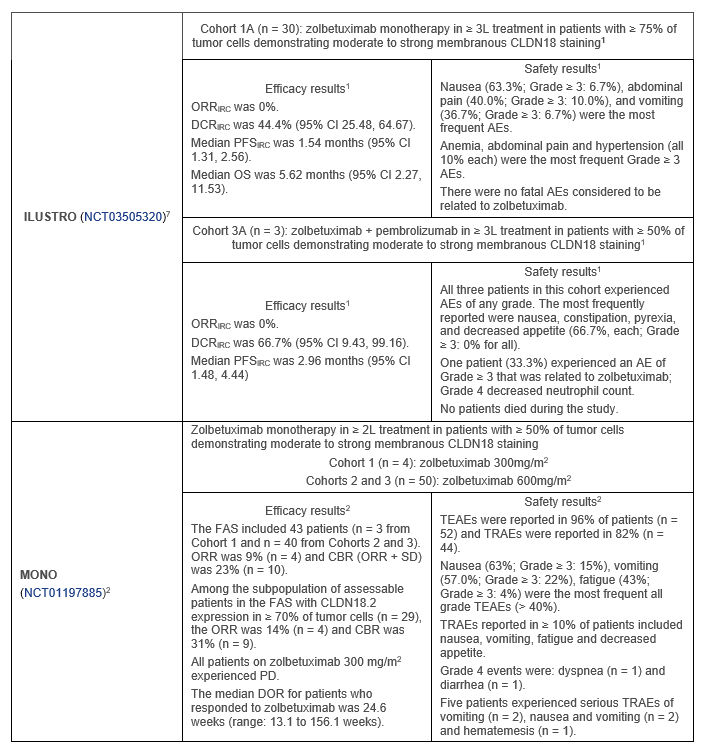
Abbreviations. 2L: second-line; 3L: third-line; AE: adverse event; CAPOX: capecitabine and oxaliplatin chemotherapy; CBR: clinical benefit rate; CI: confidence interval; CLDN18: claudin 18; CLDN18.2: claudin 18 isoform 2; CR: complete response; DCR: disease control rate; DOR: duration of response; EOX: epirubicin, oxaliplatin, capecitabine; FAS: full analysis set; G/GEJ: gastric/gastroesophageal junction; IRC: independent review committee; mFOLFOX6: modified folinic acid (or levofolinate), fluorouracil, and oxaliplatin; ORR: objective response rate; OS: overall survival; PD: progressive disease; PFS: progression-free survival; PR: partial response; SD: stable disease; TEAE: treatment emergent AE; TRAE: treatment-related AE.
Ongoing clinical trials
ZELDA (jRCTs071250038) is a Phase 2 trial evaluating the efficacy and safety of zolbetuximab in the 2L for CLDN18.2 positive advanced G/GEJ cancer patients previously treated with zolbetuximab and platinum-based chemotherapy.8,9
RAINSPOT (NCT06962137) is a Phase 2 trial evaluating whether adding zolbetuximab to paclitaxel + ramucirumab can improve patients' survival in the 2L for CLDN18.2 positive gastroesophageal cancer.10
References
1. Klempner SJ, Lee KW, Shitara K, et al. ILUSTRO: Phase 2 Multicohort Trial of Zolbetuximab in Patients with Advanced or Metastatic Claudin 18.2-Positive Gastric or Gastroesophageal Junction Adenocarcinoma. Clin. Cancer Res. 2023;29(19):3882-3891. Available at: https://doi.org/10.1158/1078-0432.ccr-23-0204.
2. Türeci O, Sahin U, Schulze-Bergkamen H, et al. A multicentre, phase IIa study of zolbetuximab as a single agent in patients with recurrent or refractory advanced adenocarcinoma of the stomach or lower oesophagus: the MONO study. Ann. Oncol. 2019;30(9):1487-1495. Available at: https://doi.org/10.1093/annonc/mdz199.
3. Sahin U, Türeci Ö, Manikhas G, et al. FAST: a randomised phase II study of zolbetuximab (IMAB362) plus EOX versus EOX alone for first-line treatment of advanced CLDN18.2-positive gastric and gastro-oesophageal adenocarcinoma. Ann. Oncol. 2021;32(5):609-619. Available at: https://doi.org/10.1016/j.annonc.2021.02.005.
4. Shitara K, Lordick F, Bang YJ, et al. Supplement to: Zolbetuximab plus mFOLFOX6 in patients with CLDN18.2-positive, HER2-negative, untreated, locally advanced unresectable or metastatic gastric or gastro-oesophageal junction adenocarcinoma (SPOTLIGHT): a multicentre, randomised, double-blind, phase 3 trial. Lancet. 2023;401(10389):1655-1668. Available at: https://doi.org/10.1016/s0140-6736(23)00620-7.
5. Shah MA, Shitara K, Ajani JA, et al. Supplement to: Zolbetuximab plus CAPOX in CLDN18.2- positive gastric or gastroesophageal junction adenocarcinoma: the randomized, phase 3 GLOW trial. Nat. Med. 2023;29(8):2133-2141. Available at: https://doi.org/10.1038/s41591-023-02465-7.
6. Data on file.
7. Electronic Citation. NIH. Clinicaltrials.gov NCT03505320. ILUSTRO. Available at: https://clinicaltrials.gov/study/NCT03505320/. Accessed 27-MAR-2026.
8. Electronic Citation. jRCT. jRCTs071250038. ZELDA. Available at: https://jrct.mhlw.go.jp/en-latest-detail/jRCTs071250038/. Accessed 27-MAR-2026.
9. Ando K, Inagaki C, Narita Y, et al. Randomized phase II trial evaluating the efficacy of zolbetuximab in the second-line for claudin18.2 positive advanced gastric/EGJ cancer patients previously treated with zolbetuximab and platinum-based chemotherapy (ZELDA trial) [oral slide presentation]. American Society of Clinical Oncology (ASCO) Gastrointestinal Cancers Symposium 2026; January 8-10, 2026. San Francisco, CA. Abstract TPS470.
10. Electronic Citation. NIH. Clinicaltrials.gov NCT06962137. RAINSPOT. Available at: https://clinicaltrials.gov/study/NCT06962137/. Accessed 27-MAR-2026.
The medical information on this website is for educational purposes only and is intended to provide scientific information about Astellas products. This information is not intended as medical advice or clinical recommendations. This website is for use only by United States residents and licensed healthcare professionals (HCPs) practicing in the United States. Product labeling may vary between countries.
Please choose an option that best describes you:
For visitors outside the United States: click here