
In SPOTLIGHT and GLOW, the incidence of nausea/vomiting was most frequent in Cycle 1 with zolbetuximab + mFOLFOX6/CAPOX and decreased in later cycles
In the combined SPOTLIGHT and GLOW analysis, nausea and/or vomiting were most common on the first infusion day of zolbetuximab and generally decreased on infusion Days 2 to 4.1
Figure 1. All occurrence of nausea or vomiting in SPOTLIGHT2
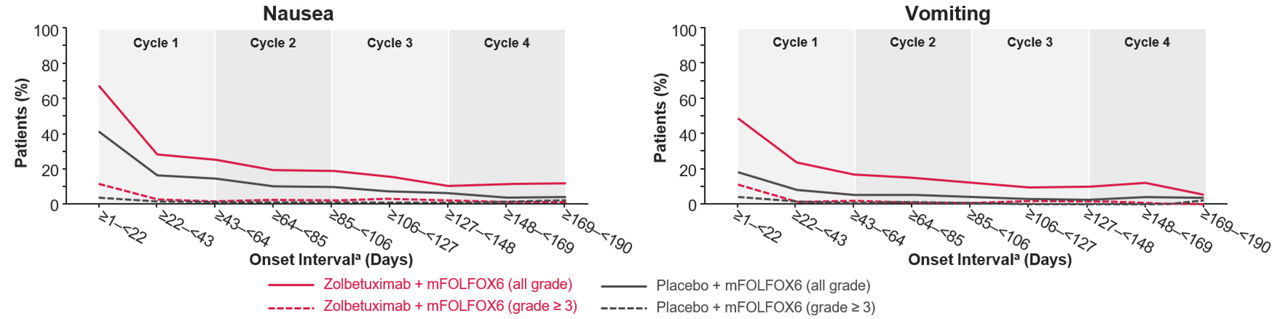
Zolbetuximab or placebo with mFOLFOX6 were administered in 42-day cycles.
aThe onset day in the onset interval was defined as the date of onset minus the date of first dose plus 1.
Abbreviations: mFOLFOX6: modified leucovorin calcium (folinic acid), fluorouracil, and oxaliplatin.
Adapted from: Supplement to Shitara et al. Lancet. 2023.
Figure 2. All occurrence of nausea or vomiting in GLOW3
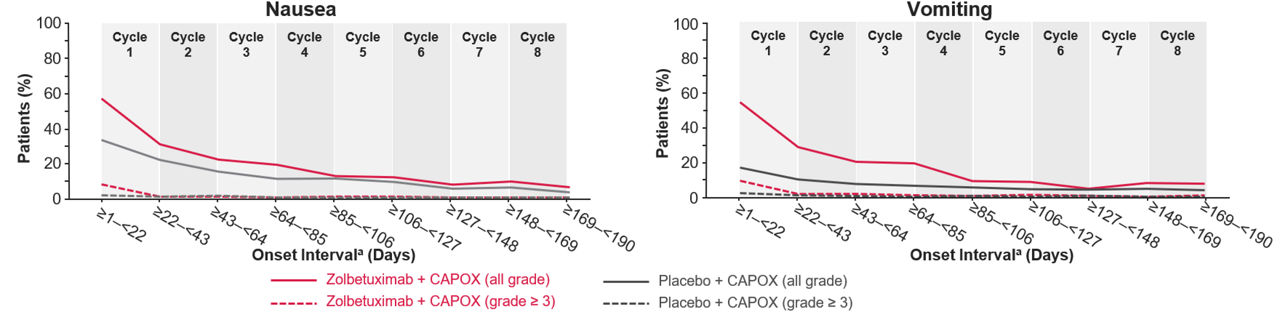
Zolbetuximab or placebo with CAPOX were administered in 21-day cycles.
aThe onset day in the onset interval was defined as the date of onset minus the date of first dose plus 1.
Abbreviations: CAPOX: capecitabine and oxaliplatin.
Adapted from: Shah et al. Nat Med. 2023.
Figure 3. Occurrences of nausea or vomiting on the day of infusion for infusions 1 to 4 in patients who received zolbetuximab or placeboa,1
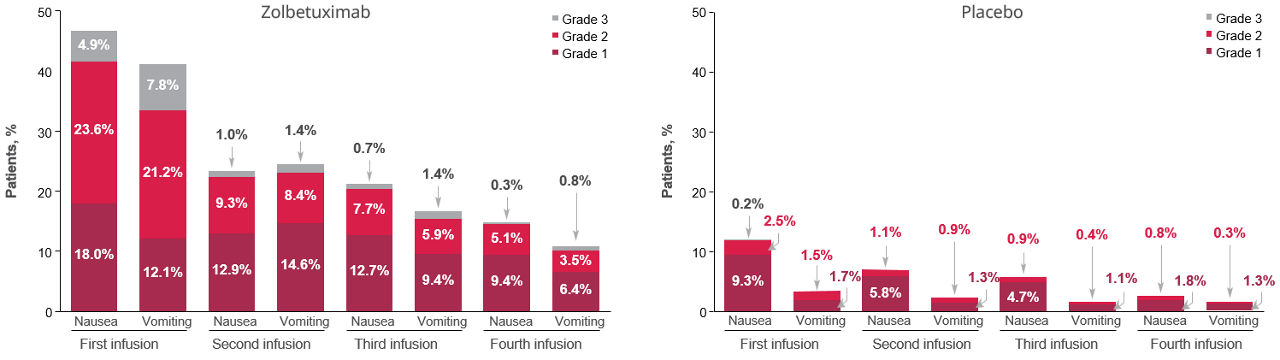
aData represent the combined analysis of SPOTLIGHT and GLOW.
Adapted from: Shitara et al. ESMO Open 2026.
Shitara K, Smith E, Lordick F, et al. Impact and effective management of nausea/vomiting on patients treated with zolbetuximab + chemotherapy: insights from the phase III SPOTLIGHT and GLOW studies. ESMO Open. 2026;epub ahead of print105931. Available at: https://doi.org/10.1016/j.esmoop.2025.105931.
Shitara K, Lordick F, Bang YJ, et al. Supplement to: Zolbetuximab plus mFOLFOX6 in patients with CLDN18.2-positive, HER2-negative, untreated, locally advanced unresectable or metastatic gastric or gastro-oesophageal junction adenocarcinoma (SPOTLIGHT): a multicentre, randomised, double-blind, phase 3 trial. Lancet. 2023;401(10389):1655-1668. Available at: https://doi.org/10.1016/s0140-6736(23)00620-7.
Shah MA, Shitara K, Ajani JA, et al. Zolbetuximab plus CAPOX in CLDN18.2-positive gastric or gastroesophageal junction adenocarcinoma: the randomized, phase 3 GLOW trial. Nat. Med. 2023;29(8):2133-2141. Available at: https://doi.org/10.1038/s41591-023-02465-7.
The medical information on this website is for educational purposes only and is intended to provide scientific information about Astellas products. This information is not intended as medical advice or clinical recommendations. This website is for use only by United States residents and licensed healthcare professionals (HCPs) practicing in the United States. Product labeling may vary between countries.
Please choose an option that best describes you:
For visitors outside the United States: click here