
There is no full analysis examining duration of nausea/vomiting in Phase 3 trials; limited post hoc data show median nausea duration 3 days and vomiting 1 day
A post hoc integrated analysis of Phase 2 (FAST, ILUSTRO) and Phase 3 (SPOTLIGHT, GLOW) studies was conducted comparing zolbetuximab + EOX/mFOLFOX6/CAPOX (n = 631) with EOX or placebo + mFOLFOX6/CAPOX (n = 611).1
Caution should be used in interpreting the results of this post-hoc analysis due to limitations in data collection.1 If nausea or vomiting changed grade, there was no adverse event identification to link it and the duration included in the analysis was not the full duration of the event.
Table 1. Duration of nausea/vomiting events in the integrated FAST/ILUSTRO/SPOTLIGHT/GLOW studies (safety analysis set)1
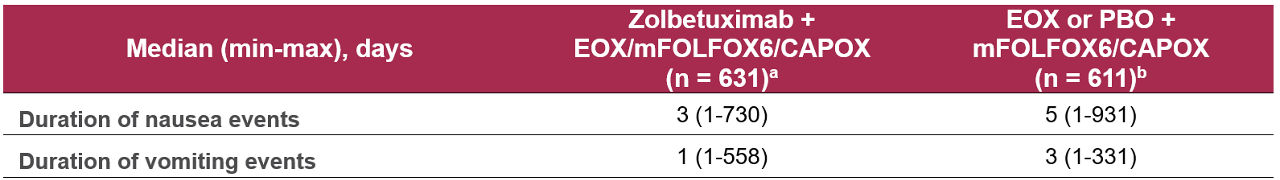
aIncluding patients who received zolbetuximab 800/600 mg/m2 + EOX (FAST), mFOLFOX6 (ILUSTRO or SPOTLIGHT) or CAPOX (GLOW).
bIncluding patients who received EOX alone (FAST), Placebo + mFOLFOX6 (SPOTLIGHT) or CAPOX (GLOW).
Abbreviations: CAPOX: capecitabine and oxaliplatin; EOX: epirubicin, oxaliplatin, capecitabine; max: maximum; min: minimum; mFOLFOX6: modified 5-fluorouracil, leucovorin (or folinic acid) and oxaliplatin; PBO: placebo.
Data on File.
The medical information on this website is for educational purposes only and is intended to provide scientific information about Astellas products. This information is not intended as medical advice or clinical recommendations. This website is for use only by United States residents and licensed healthcare professionals (HCPs) practicing in the United States. Product labeling may vary between countries.
Please choose an option that best describes you:
For visitors outside the United States: click here