
ACP 2 mg reduced the progression to driving ineligibility compared with sham, particularly when BCVA is ≥ 75 letters.
A lower proportion of ACP-treated eyes treated with ACP 2 mg progressed to driving ineligibility over 12 months when compared with sham.1 Pooled 24-month data suggest patients with geographic atrophy (GA) receiving ACP 2 mg may be less likely than those receiving sham to progress to driving restrictions or lose driving eligibility if they are treated when best corrected visual acuity (BCVA) is ≥ 75 letters.2
Two separate sub-analyses were conducted with different methodological approaches:
Danzig et al. sub-analysis:1
Figure 1. Proportion of patients with BCVA loss below driving eligibility threshold1
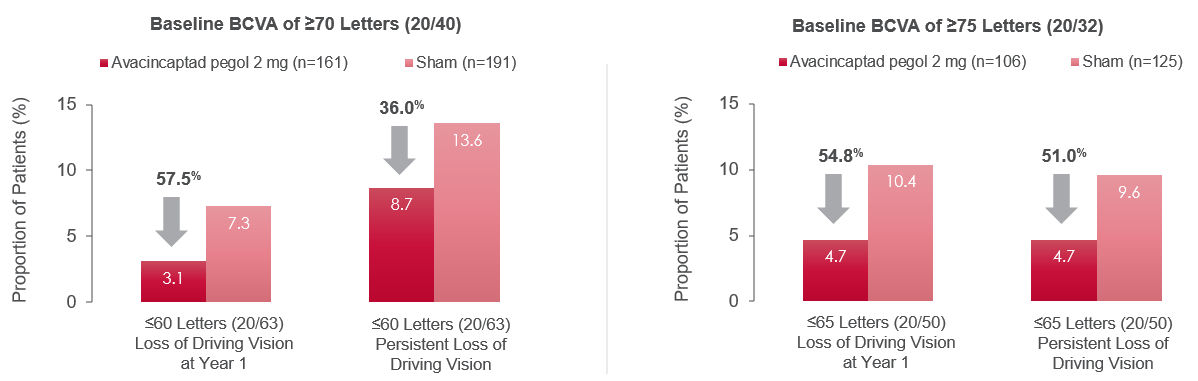
“Loss” defined as BCVA of ≤ 60 letters (cutoff 1) or ≤ 65 letters (cutoff 2) at Month 12 (left graphs).
“Persistent loss” defined as BCVA of ≤ 60 letters (cutoff 1) or ≤ 65 letters (cutoff 2) at two consecutive postbaseline visits (right graphs).
Adapted from: Danzig CJ, Ophthalmol Retina, 2024.
Hariprasad et al. subanalysis:2
Figure 2. Definitions of driving eligibility.
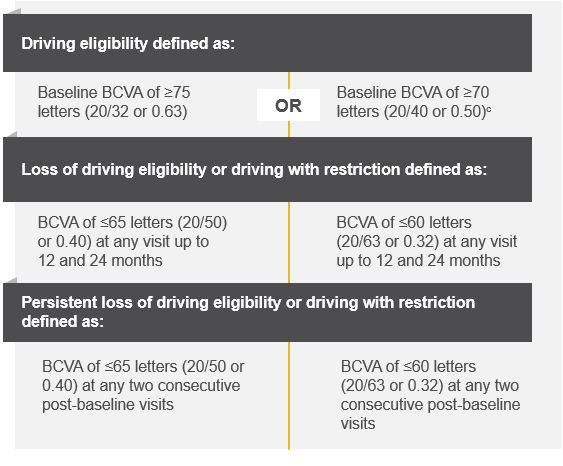
Adapted from: Hariprasad SM, American Society of Retina Specialists (ASRS), 2024.
Figure 3. Proportion of patients with BCVA loss below driving eligibility threshold up to 12 monthsa, 2
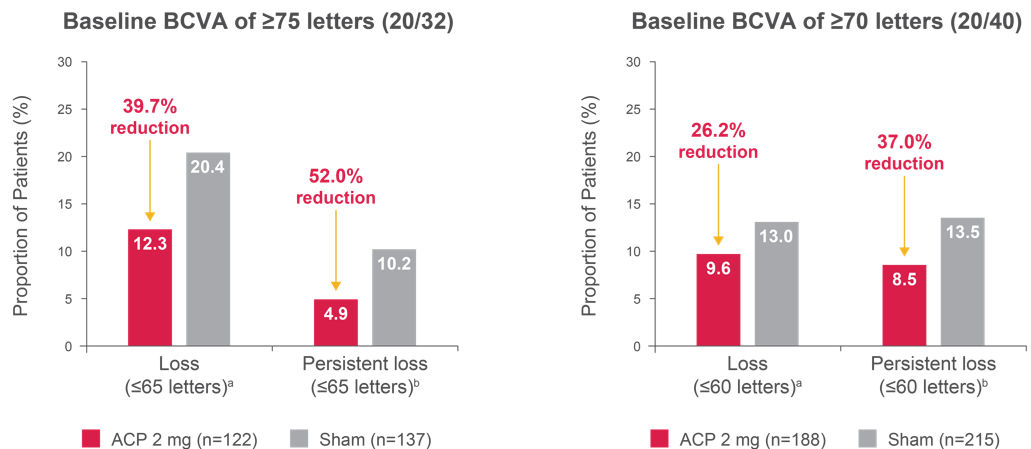
Left graph comparisons: Loss, p = 0.0947; persistent loss, p = 0.1608. Right graph comparisons: Loss, p = 0.3463; persistent loss, p = 0.1531.
a“Loss” defined as BCVA of ≤ 65 letters (left graph) or ≤ 60 letters (right graph) at any postbaseline visit.
b“Persistent loss” defined as BCVA of ≤ 65 letters (left graph) or ≤ 60 letters (right graph) at two consecutive postbaseline visits.
Adapted from: Hariprasad SM, ASRS, 2024.
Figure 4. Proportion of patients with BCVA loss below driving eligibility threshold up to 24 monthsa, 2
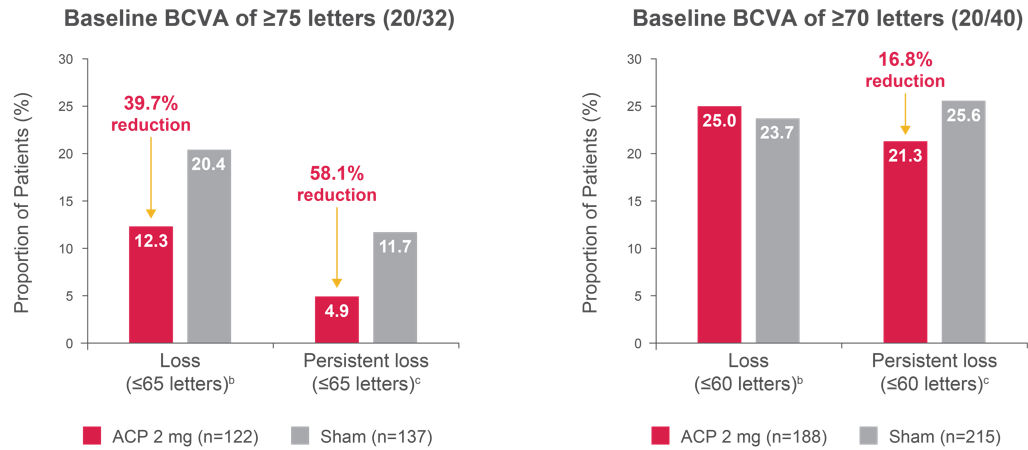
Left graph comparisons: Loss, p = 0.0947; persistent loss, p = 0.0726. Right graph comparisons: Loss, p = 0.8162; persistent loss, p = 0.3473.
aThere was no change in the proportion of patients with “loss of driving eligibility” in the baseline BCVA of ≥ 70 letters group.
b“Loss” defined as BCVA of ≤ 65 letters (left graph) or ≤ 60 letters (right graph) at any postbaseline visit.
c“Persistent loss” defined as BCVA of ≤ 65 letters (left graph) or ≤ 60 letters (right graph) at two consecutive postbaseline visits.
Adapted from: Hariprasad SM, ASRS, 2024.
The medical information on this website is for educational purposes only and is intended to provide scientific information about Astellas products. This information is not intended as medical advice or clinical recommendations. This website is for use only by United States residents and licensed healthcare professionals (HCPs) practicing in the United States. Product labeling may vary between countries.
Please choose an option that best describes you:
For visitors outside the United States: click here